Medically reviewed content. Written by Jonathan Bailor, Founder of SANESolution. Updated March 19, 2026. Part of the Consumer Health Guide research library.
Postbiotics and butyrate may offer more consistent, safer, and more effective gut support than traditional probiotics for many people. While probiotics have dominated the gut health conversation for over two decades, the science now points to a simpler truth: much of what makes probiotics useful comes from what they produce, not the live bacteria themselves. Postbiotics, especially the short-chain fatty acid butyrate, deliver those benefits directly, without the unpredictability of live organisms. (see gut microbiome research) (see probiotic safety)
I’ve spent years reviewing the clinical literature on gut health interventions, and the shift toward postbiotics is one of the most significant changes I’ve seen. The 2021 ISAPP (International Scientific Association for Probiotics and Prebiotics) consensus panel, published in Nature Reviews Gastroenterology and Hepatology, formally defined postbiotics as “a preparation of inanimate microorganisms and/or their components that confers a health benefit on the host.” That formal recognition marked a turning point for how researchers and clinicians think about gut supplementation.
Prebiotics, Probiotics, and Postbiotics: What’s the Actual Difference?
Before comparing these approaches, you need to understand what each one does and how they relate to each other.
Prebiotics are non-digestible fibers (like inulin, fructo-oligosaccharides, and resistant starch) that feed your existing gut bacteria. Think of them as fertilizer for the microbes already living in your colon. Foods high in prebiotics include garlic, onions, leeks, asparagus, bananas, and oats.
Probiotics are live bacteria and yeasts, typically from the Lactobacillus and Bifidobacterium genera, taken as supplements or found in fermented foods like yogurt, kefir, kimchi, and sauerkraut. The idea is that adding “good” bacteria to your gut improves the microbial balance. For more on feeding your gut well, see our guide on the best foods for gut health.
Postbiotics are the metabolic byproducts, cell fragments, and bioactive compounds that bacteria produce during fermentation. They include short-chain fatty acids (butyrate, propionate, acetate), enzymes, peptides, cell wall fragments, and organic acids. The key difference: postbiotics are not alive. They don’t need to survive your stomach acid, colonize your intestines, or compete with your existing microbiome.
Here’s the part most supplement companies won’t tell you: a growing body of research suggests that probiotics’ benefits come primarily from the metabolites they produce (postbiotics), not from the live bacteria themselves. Multiple reviews have noted that conclusive evidence of probiotic colonization in the gut mucosa is still lacking, even after years of research.
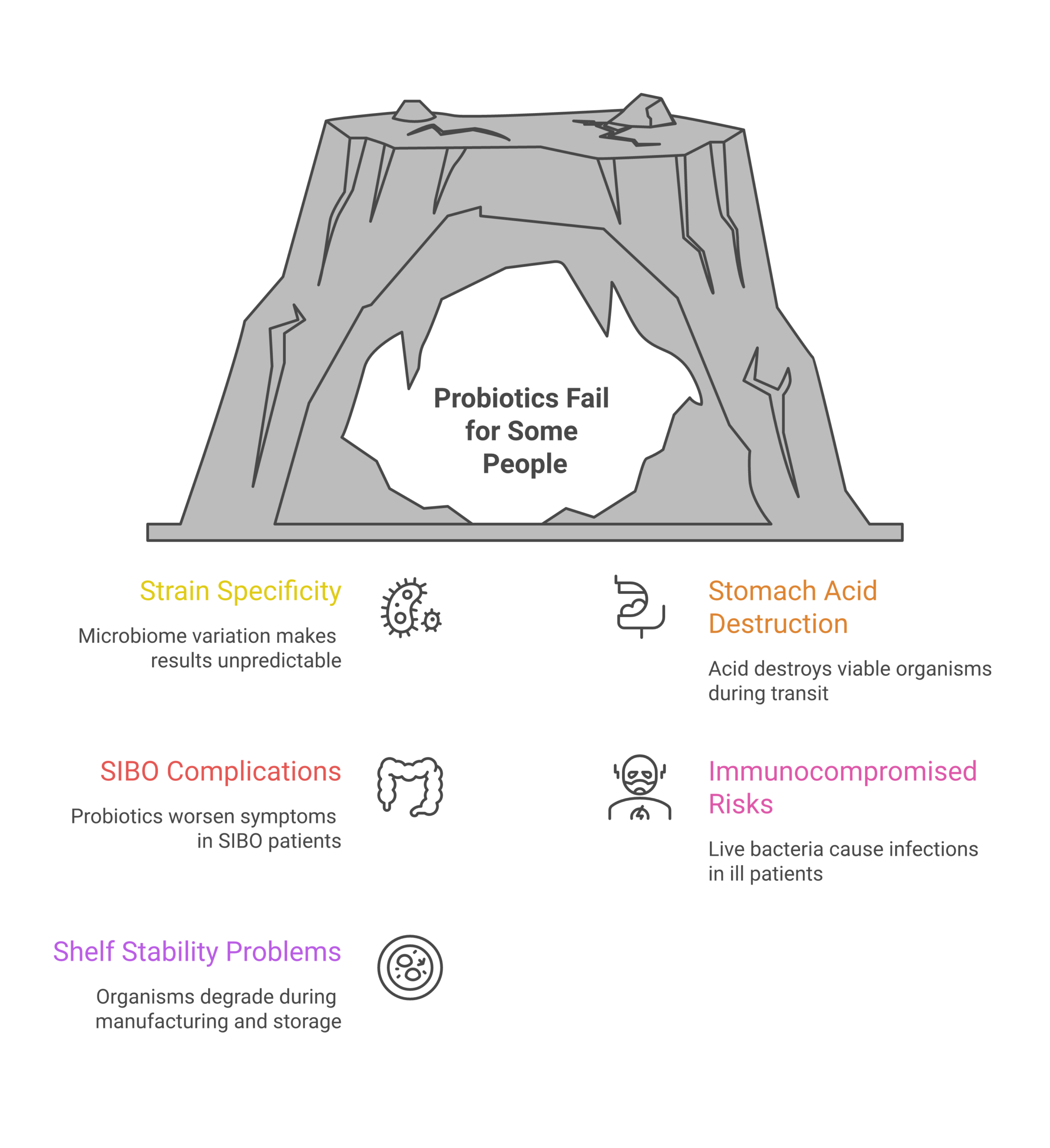
Why Probiotics Fail for Some People
Probiotics are not universally effective. In my clinical research review, I’ve identified five specific reasons why probiotics underperform or cause problems for certain individuals.
Gut health also influences your skin through what researchers call the gut-skin axis. Learn which topical ingredients complement an inside-out approach in our dark spot remover ingredients.
Your gut and brain are connected through the vagus nerve and the gut-brain axis. If you are also experiencing nerve discomfort, see our nerve supplement guide for ingredients backed by clinical research.
1. Strain Specificity and Individual Variation
Your gut microbiome is as unique as your fingerprint. A probiotic strain that helps one person may do nothing for another. Your existing microbial composition, diet, medication history, and genetics all influence whether a particular strain can even survive in your gut, let alone provide benefits. Most commercial probiotics contain a handful of strains, but there are over 1,000 bacterial species in the human gut.
2. Stomach Acid Destruction
Live bacteria must survive the harsh acidic environment of your stomach (pH 1.5-3.5) to reach the intestines where they’re needed. Many strains don’t make it. A study published in the European Journal of Pharmaceutics and Biopharmaceutics found that uncoated probiotic capsules can lose 60-80% of viable organisms during gastric transit. You’re often paying for bacteria that never arrive alive.
3. SIBO Complications
For the estimated 6-15% of healthy individuals (and up to 80% of IBS patients) who have Small Intestinal Bacterial Overgrowth, probiotics can make things worse. Introducing additional bacteria into an already overpopulated small intestine increases bloating, gas, and abdominal pain. A clinical study found that probiotic users were significantly more likely to have positive methane breath tests (93.6%) compared to non-users (64.3%) in patients presenting with suspected SIBO symptoms.
4. Risks for Immunocompromised Individuals
Live bacterial supplements carry real infection risks for people with weakened immune systems. Case reports in medical literature document probiotic-related bacteremia and fungemia in critically ill and immunocompromised patients. People undergoing chemotherapy, taking immunosuppressive medications, or living with conditions like HIV need to be especially cautious with live bacterial supplements. Postbiotics, by contrast, contain no live organisms and carry virtually no infection risk.
5. Shelf Stability and Potency Loss
Probiotics are living organisms that need specific temperature and humidity conditions. Many products lose 50% or more of their CFU (colony forming units) count during manufacturing, shipping, and storage. The number on the label often doesn’t match what’s actually alive in the capsule by the time you take it. Independent lab testing by ConsumerLab has repeatedly found probiotic supplements that contain far fewer viable organisms than claimed.
What Makes Postbiotics Different (and Often Better)
Postbiotics skip the middleman. Instead of hoping live bacteria survive, colonize, and then produce beneficial compounds, postbiotics deliver those compounds directly.
Shelf Stability
Because postbiotics are not alive, they don’t degrade at room temperature. They don’t need refrigeration. They maintain consistent potency from manufacturing through the last capsule in the bottle. For consumers, this means what you buy is what you get, every time.
Consistent Dosing
With probiotics, the amount of beneficial metabolites your body actually receives depends on survival rates, colonization success, and fermentation activity, all of which vary day to day. Postbiotic supplements deliver a precise, measurable dose of active compounds with every serving. No guesswork.
Safety for Sensitive Populations
The 2021 ISAPP consensus panel specifically noted the favorable safety profile of postbiotics in populations where live bacteria carry risks. For people who are immunocompromised, elderly, critically ill, or dealing with SIBO, postbiotics provide gut support without introducing live organisms that could cause infections or worsen bacterial overgrowth.
No Colonization Required
Probiotics need to establish themselves in your existing microbial ecosystem, a process that often fails. Postbiotics work regardless of your current microbiome composition. They interact directly with intestinal epithelial cells, immune cells, and the gut barrier without needing to compete for space against trillions of resident bacteria.
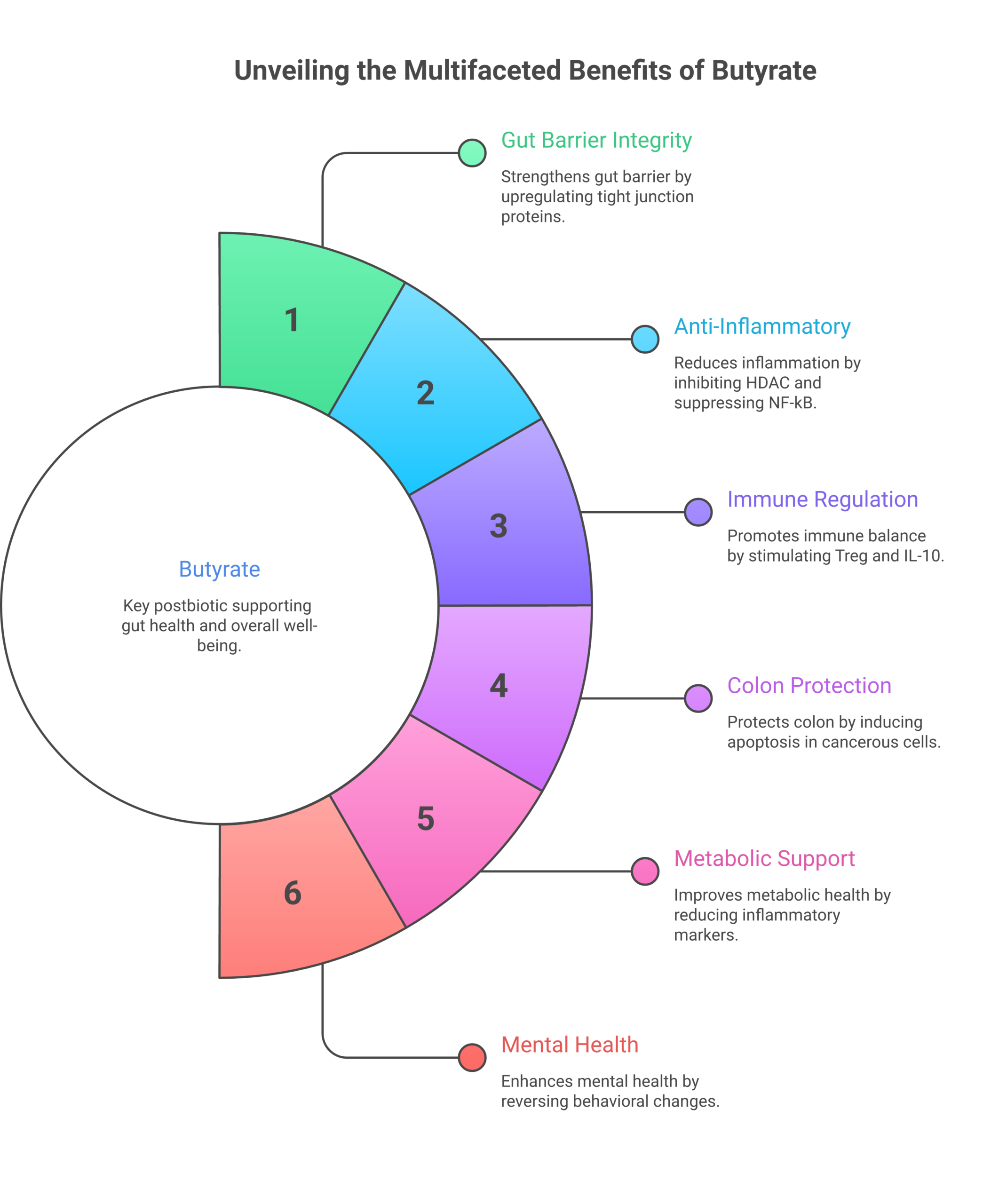
Butyrate: The Star Postbiotic
Among all postbiotic compounds, butyrate stands out. It’s a four-carbon short-chain fatty acid produced when gut bacteria ferment dietary fiber, and it serves as the primary energy source for colonocytes (the cells lining your colon). Without adequate butyrate, your colon lining weakens, inflammation increases, and gut barrier function breaks down.
What Butyrate Does in Your Body
The research on butyrate is extensive. Here’s what the clinical evidence shows:
- Gut barrier integrity: Butyrate strengthens tight junctions between intestinal cells. A 2023 review in Advances in Nutrition confirmed that butyrate upregulates tight junction proteins (claudin-1, occludin, ZO-1), directly reducing intestinal permeability, often called “leaky gut.”
- Anti-inflammatory action: Butyrate acts as a histone deacetylase (HDAC) inhibitor, suppressing NF-kB activation and reducing pro-inflammatory cytokines like TNF-alpha and IL-6. A 2025 randomized controlled trial in ulcerative colitis patients found that butyrate supplementation significantly reduced disease severity and improved psychological symptoms.
- Immune regulation: Butyrate promotes the differentiation of regulatory T cells (Tregs), which help prevent autoimmune reactions and excessive inflammation. It also stimulates IL-10 production, a key anti-inflammatory cytokine.
- Colon cancer protection: Butyrate induces apoptosis (programmed cell death) in cancerous colonocytes while promoting healthy cell growth. This “butyrate paradox,” where it fuels normal cells but kills cancerous ones, has been documented in multiple in vitro and animal studies.
- Metabolic benefits: A study giving 4 grams of sodium butyrate daily for 4 weeks to obese individuals found measurable reductions in inflammatory markers and improvements in trained immunity in monocytes.
- Mental health connections: Clinical data shows that fecal butyrate concentrations are lower in patients with depression compared to healthy controls. Animal studies demonstrate that butyrate supplementation reverses behavioral changes associated with depression, including low energy and social withdrawal.
The Tributyrin Advantage
Not all butyrate supplements are equal. Standard sodium butyrate has poor bioavailability because it’s rapidly absorbed in the upper GI tract and largely destroyed before reaching the colon, where it’s most needed. If you’re considering a postbiotic supplement, our guide on what to look for in a postbiotic supplement covers the key quality markers.
Tributyrin, a triglyceride form of butyrate (three butyrate molecules attached to a glycerol backbone), solves this problem. A 2025 study published in Frontiers in Nutrition tested CoreBiome tributyrin in an in vitro simulation of the human GI tract and found that over 50% of tributyrin remained stable during upper GI transit. Between 40.9% and 48.7% was hydrolyzed to butyrate in the small intestine, and the remaining 51.3-59.1% reached the colon intact, where it was gradually released. For a deeper look at why tributyrin outperforms other butyrate forms, read our article on why CoreBiome is the best patented form of butyrate.
That same study found tributyrin supplementation increased beneficial bacterial species, enhanced intestinal barrier function, boosted IL-10 (anti-inflammatory) levels, and decreased TNF-alpha (pro-inflammatory) levels in colonic fermentation models.
Probiotics vs. Postbiotics: Head-to-Head Comparison
| Feature | Probiotics | Postbiotics (Butyrate/Tributyrin) |
|---|---|---|
| Contains live organisms | Yes | No |
| Requires refrigeration | Often yes | No |
| Survives stomach acid | Variable (20-40% loss common) | Yes (over 50% reaches colon intact) |
| Colonization needed | Yes, and often fails | No |
| Consistent dosing | Difficult to guarantee | Precise and repeatable |
| Safe for immunocompromised | Caution required (infection risk) | Yes (no live organisms) |
| Safe with SIBO | May worsen symptoms | Generally well tolerated |
| Shelf life | Degrades over time | Stable 12+ months at 25C |
| Works regardless of microbiome | No (depends on individual gut) | Yes |
| Direct anti-inflammatory action | Indirect (via metabolite production) | Direct (HDAC inhibition, NF-kB suppression) |
When Probiotics ARE the Better Choice
This isn’t an either/or situation. Probiotics still have clear advantages in certain contexts.
After antibiotic treatment: Antibiotics wipe out bacterial populations. In this scenario, repopulating with live organisms through probiotics (especially Saccharomyces boulardii and specific Lactobacillus strains) has strong evidence for preventing antibiotic-associated diarrhea and C. difficile infection. A Cochrane review found probiotics reduced the risk of C. difficile-associated diarrhea by about 60%.
Infant gut development: Colonizing the infant gut with beneficial bacteria during the first 1,000 days of life shapes long-term immune function. Probiotic supplementation in this window has different goals than adult supplementation, and live organisms are appropriate here.
Fermented food consumption: Whole fermented foods (yogurt, kefir, kimchi, sauerkraut) provide live bacteria alongside prebiotics, postbiotics, vitamins, and enzymes in a food matrix that supports absorption. A 2021 Stanford study published in Cell found that a high-fermented-food diet increased microbiome diversity and decreased inflammatory markers over 10 weeks. Eating fermented foods is different from taking probiotic capsules.
Well-studied strain-specific conditions: Certain probiotic strains have strong clinical evidence for specific conditions, such as Lactobacillus rhamnosus GG for pediatric diarrhea or VSL#3 for ulcerative colitis maintenance. When a well-studied strain matches a specific condition, probiotics can be the right tool.
The Synbiotic Approach: Why Both Together May Be Best
The most forward-thinking approach to gut health doesn’t choose between probiotics and postbiotics. It uses both strategically.
A synbiotic approach combines prebiotics (fiber to feed existing bacteria), targeted probiotics (when indicated), and postbiotics like tributyrin to cover all bases. This way, you’re feeding your existing microbiome, supporting it with specific strains when needed, and directly supplying the beneficial compounds your gut lining requires.
In my experience reviewing the gut health literature, the people who see the best outcomes are those who combine a high-fiber diet rich in prebiotic foods with targeted postbiotic supplementation (specifically tributyrin-based butyrate). Probiotics can be added when there’s a specific clinical reason, like post-antibiotic recovery or a condition where a well-studied strain has demonstrated clear benefits.
To see how real users have experienced postbiotic supplementation, check out our verified postbiotic reviews and gut health product reviews.
How to Choose the Right Postbiotic Supplement
If you’re considering adding a postbiotic to your routine, here’s what matters:
- Look for tributyrin, not sodium butyrate. Tributyrin delivers butyrate to the colon where it’s needed. Sodium butyrate is largely absorbed before reaching the lower GI tract.
- Check for patented, clinically tested ingredients. CoreBiome is the most-studied tributyrin ingredient, with published research on bioavailability and gut barrier function.
- Verify third-party testing. Any supplement should have independent testing for purity and potency, especially in the postbiotic category where regulation is still catching up.
- Dosing. Most clinical studies use 300-600 mg of tributyrin daily. Check that the supplement provides this range per serving.
- Avoid proprietary blends that hide individual ingredient amounts. You need to know exactly how much tributyrin you’re getting.
Browse all of our gut health supplements reviews and guides for more research-backed recommendations.
Frequently Asked Questions
Are postbiotics better than probiotics?
For many people, yes. Postbiotics provide more consistent dosing, better shelf stability, and carry no risk of bacterial infection. They work without needing to colonize your gut. However, probiotics still have value after antibiotic use and for specific clinical conditions where particular strains have strong evidence.
What is butyrate and why is it important for gut health?
Butyrate is a short-chain fatty acid that serves as the primary fuel source for the cells lining your colon. It strengthens the gut barrier, reduces inflammation through HDAC inhibition, supports immune regulation, and has been linked to reduced colon cancer risk. Low butyrate levels are associated with inflammatory bowel disease, metabolic disorders, and depression.
Can I take postbiotics and probiotics together?
Yes. Taking both is sometimes called a “synbiotic” approach (though technically, synbiotics combine probiotics with prebiotics). There are no known negative interactions between postbiotics like tributyrin and probiotic supplements. Many clinicians now recommend both for patients with significant gut dysfunction.
Are postbiotics safe for people with weakened immune systems?
Postbiotics are generally considered safe for immunocompromised individuals because they contain no live organisms. The 2021 ISAPP consensus statement specifically noted the favorable safety profile of postbiotics in sensitive populations. Probiotics, by contrast, carry documented risks of bacteremia and fungemia in critically ill and immunocompromised patients.
What is tributyrin and how is it different from regular butyrate supplements?
Tributyrin is a triglyceride form of butyrate, consisting of three butyrate molecules bound to a glycerol backbone. Unlike sodium butyrate, which is rapidly absorbed and degraded in the upper GI tract, over 50% of tributyrin survives gastric transit and delivers butyrate directly to the colon. CoreBiome is the most widely studied patented tributyrin ingredient.
Do postbiotics help with leaky gut?
Clinical evidence supports this. Butyrate (the primary postbiotic studied for intestinal permeability) upregulates tight junction proteins including claudin-1, occludin, and ZO-1. These proteins seal the gaps between intestinal cells, directly addressing the mechanism behind increased intestinal permeability. Tributyrin-based postbiotics deliver butyrate to the colon where these tight junctions are most critical.
How long do postbiotics take to work?
Most people report noticing changes in digestive comfort within 1-2 weeks of consistent tributyrin supplementation. Measurable changes in gut barrier function and inflammatory markers typically appear within 4-6 weeks in clinical studies. Unlike probiotics, which require time for colonization (if it occurs at all), postbiotics begin interacting with intestinal cells immediately upon reaching the gut.
Can I get enough postbiotics from food alone?
Your gut bacteria naturally produce postbiotics, including butyrate, when they ferment dietary fiber. Eating a diet rich in prebiotic fibers (garlic, onions, leeks, asparagus, oats, resistant starch) increases natural butyrate production. However, people with dysbiosis, low-fiber diets, or conditions that disrupt the microbiome may not produce enough butyrate on their own. Supplementation with tributyrin can fill that gap.
This article is for informational purposes only and does not constitute medical advice. Consult your healthcare provider before starting any new supplement, especially if you have existing health conditions or take medications.

