By Jonathan Bailor, Founder of SANESolution. Part of the Consumer Health Guide research library.
Most over-the-counter dark spot removers are safe when used as directed, but safety depends entirely on the active ingredients, their concentrations, and your skin type. Ingredients like vitamin C, niacinamide, and azelaic acid have strong safety records supported by decades of clinical research. Others, like hydroquinone and certain retinoids, carry real risks that range from skin irritation to, in rare cases, permanent discoloration or harm during pregnancy. The short answer: OTC dark spot products with well-studied ingredients are generally safe for most adults. But you need to know what you’re putting on your skin, and I’ll walk you through every ingredient, every risk, and every red flag below.
Why Dark Spot Remover Safety Matters More Than You Think
Dark spots (also called hyperpigmentation) affect millions of people across all skin tones, and the global skin-lightening market was valued at over $8.6 billion in 2024, according to Grand View Research. That’s a lot of products, a lot of marketing claims, and a lot of room for confusion about what’s actually safe.
I’ve spent years researching skin health from a whole-body perspective, and one thing I’ve learned is that “natural” doesn’t always mean safe, and “clinical-strength” doesn’t always mean dangerous. The truth sits somewhere in the middle, and it comes down to specific ingredients at specific concentrations.
For anyone with hyperpigmentation, whether from sun damage, acne scarring, or hormonal changes like melasma, choosing a safe dark spot remover isn’t optional. It’s a health decision. YMYL topics like this one demand accuracy, and that’s what I’m giving you here.
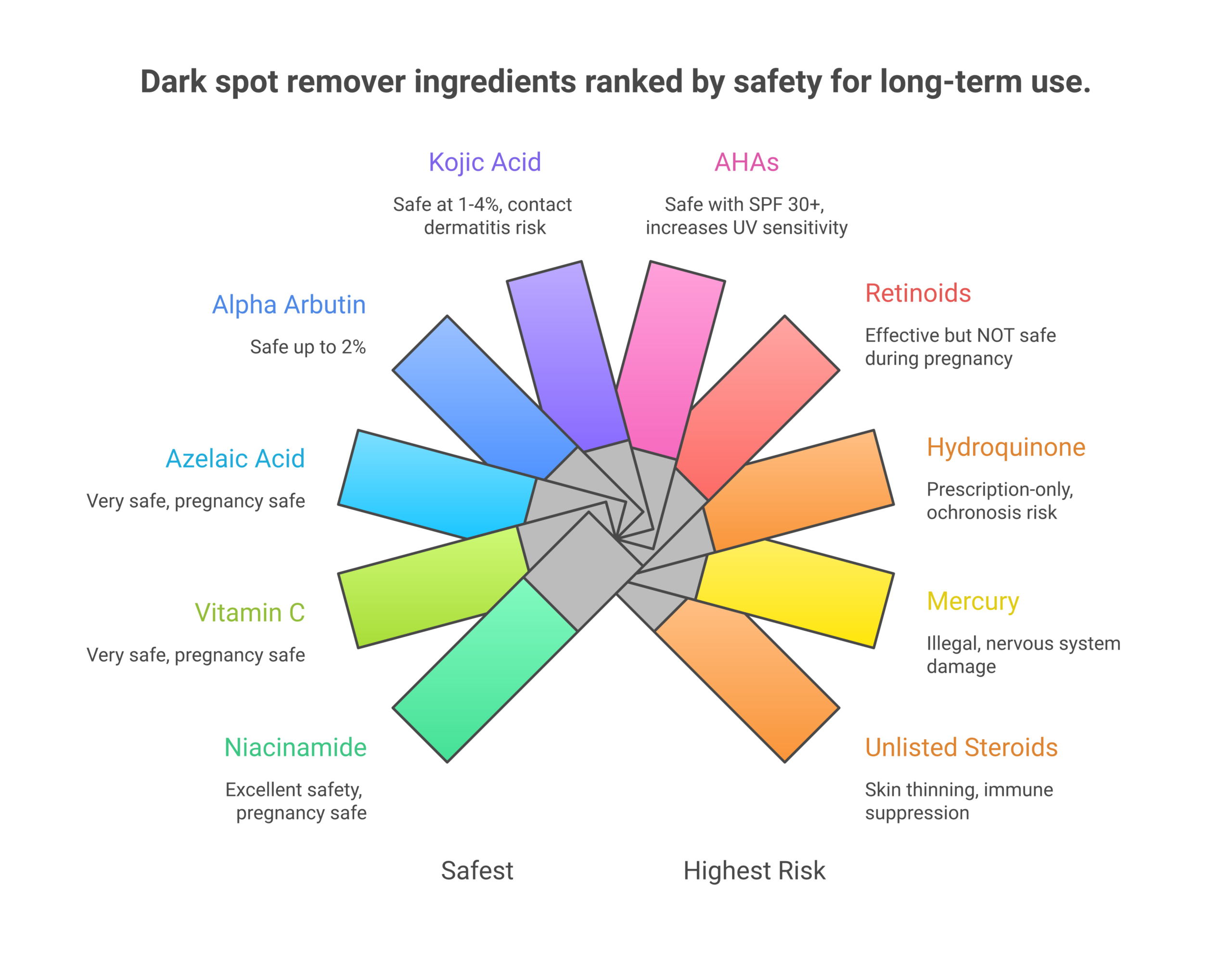
Ingredient-by-Ingredient Safety Breakdown
Not all dark spot removers are created equal. The safety profile of any product depends on what’s in it. Here’s what the clinical evidence actually says about the most common active ingredients.
Skin health starts from the inside. Gut inflammation can worsen hyperpigmentation and slow healing. Our postbiotic supplement guide explains which supplements support the gut-skin connection.
Vitamin C (Ascorbic Acid and Derivatives)
Safety rating: Very safe for most people.
Vitamin C is one of the most widely studied ingredients in dark spot removers. It works by inhibiting tyrosinase, the enzyme responsible for melanin production. Concentrations between 10% and 20% are standard in OTC serums.
Side effects are minimal. Some people with sensitive skin report mild stinging or tingling upon application, especially with L-ascorbic acid at higher concentrations. The bigger challenge with vitamin C is stability, not safety. Ascorbic acid oxidizes quickly when exposed to light and air, turning the product brown and less effective. Derivatives like sodium ascorbyl phosphate and magnesium ascorbyl phosphate are more stable but slightly less potent.
A 2019 review published in the Journal of Clinical and Aesthetic Dermatology confirmed that topical vitamin C has an excellent safety profile at concentrations up to 20%, with irritation risk increasing above that threshold.
Niacinamide (Vitamin B3)
Safety rating: Excellent. One of the safest active ingredients available.
Niacinamide has one of the best safety records of any dark spot treatment ingredient. It works by blocking the transfer of melanosomes (pigment-containing structures) from melanocytes to keratinocytes. A 2011 study in the British Journal of Dermatology found that 5% niacinamide significantly reduced hyperpigmentation after 8 weeks with no serious adverse events reported.
Concentrations between 2% and 10% are typical in OTC products. Irritation is rare, even in sensitive skin. Niacinamide is also safe during pregnancy and breastfeeding, which makes it one of the few dark spot ingredients that pregnant women can use without concern.
Hydroquinone: The Controversial One
Safety rating: Effective but carries real risks. Prescription-only in the U.S. since 2020.
Hydroquinone was once the gold standard for treating dark spots. It works by inhibiting tyrosinase more aggressively than most other ingredients. But its safety profile has been a source of ongoing debate for over two decades.
Here’s the timeline of key regulatory actions:
- 2006: The FDA proposed a rule stating that OTC hydroquinone products were not “generally recognized as safe and effective” (GRASE). The proposal cited evidence of carcinogenicity in rats and mice, plus reports of ochronosis, a blue-black discoloration of the skin caused by long-term use at concentrations as low as 1-2%.
- 2006-2020: The proposed rule languished without a final decision for 14 years.
- 2020: The CARES Act (Coronavirus Aid, Relief, and Economic Security Act) reformed OTC drug regulation. Under the new rules, any drug without a final monograph and not considered GRASE needed an approved New Drug Application (NDA) to remain on the market. This effectively removed OTC hydroquinone products from legal sale as of September 23, 2020.
- Current status: Tri-Luma (a combination of hydroquinone, tretinoin, and fluocinolone) is the only FDA-approved prescription product containing hydroquinone, approved specifically for short-term treatment of moderate-to-severe melasma.
The key risks with hydroquinone include:
- Ochronosis: Paradoxical permanent darkening of the skin. Documented at concentrations as low as 1-2% with prolonged use.
- Rebound hyperpigmentation: Dark spots can return worse than before after stopping the product.
- Cancer concerns: Animal studies showed evidence of carcinogenicity, though human data remains limited.
- Skin irritation: Redness, peeling, and burning, especially at concentrations above 4%.
If your dermatologist prescribes hydroquinone, follow their guidance on duration (typically no more than 3-4 months of continuous use) and always use sunscreen alongside it.
Retinoids (Retinol, Tretinoin, Adapalene)
Safety rating: Effective but requires caution. Not safe during pregnancy.
Retinoids speed up cell turnover, which gradually pushes pigmented cells to the surface and replaces them with new, evenly pigmented cells. OTC retinol is weaker than prescription tretinoin, but both carry similar side effect profiles at different intensities.
Common side effects include dryness, flaking, redness, and significant sun sensitivity. These effects usually improve after 4-6 weeks as your skin adjusts, a period dermatologists call “retinization.”
The pregnancy issue is non-negotiable. Both oral and topical retinoids are classified as teratogens, meaning they can cause birth defects. The American Academy of Dermatology (AAD) advises against all retinoid use during pregnancy and breastfeeding. Four published case reports have documented birth defects consistent with retinoid embryopathy from topical tretinoin use. If you’re pregnant, planning to become pregnant, or breastfeeding, avoid retinoids entirely and switch to pregnancy-safe alternatives like azelaic acid, vitamin C, or niacinamide.
Alpha Hydroxy Acids (AHAs): Glycolic Acid, Lactic Acid, Mandelic Acid
Safety rating: Safe at OTC concentrations with proper sun protection.
AHAs exfoliate the skin’s surface, which helps fade dark spots by removing pigmented dead skin cells. Glycolic acid (from sugarcane) has the smallest molecular size and penetrates most deeply. Lactic acid is gentler and also provides hydration. Mandelic acid, with its larger molecule, is often recommended for sensitive or melanin-rich skin because it penetrates more slowly and causes less irritation.
The primary safety concern with AHAs is increased sun sensitivity. A 2003 FDA-funded study found that glycolic acid at 10% concentration increased UV sensitivity by 18%. This means daily broad-spectrum sunscreen (SPF 30 or higher) is absolutely required when using AHA-based dark spot treatments. Without it, you risk making your dark spots worse.
OTC products typically contain AHAs at 5-12%. Professional chemical peels go higher (20-70%), and those carry greater risks including burns, scarring, and post-inflammatory hyperpigmentation, especially on darker skin tones.
Azelaic Acid
Safety rating: Very safe. One of the few ingredients safe during pregnancy.
Azelaic acid inhibits tyrosinase and also has anti-inflammatory and antibacterial properties. It’s FDA-approved at 15% (Finacea) for rosacea and at 20% (Azelex) for acne, but dermatologists frequently prescribe it off-label for hyperpigmentation.
It’s classified as Pregnancy Category B by the FDA, meaning animal studies have shown no fetal risk and it’s generally considered safe for pregnant and nursing women. This makes azelaic acid a go-to recommendation for women dealing with melasma during pregnancy.
Side effects are mild: temporary burning, stinging, or tingling in the first few weeks. These typically resolve on their own.
Kojic Acid
Safety rating: Generally safe at standard concentrations (1-4%).
Derived from fungi during fermentation of rice (specifically Aspergillus oryzae), kojic acid inhibits tyrosinase similarly to hydroquinone but with a milder effect. It’s often combined with other brightening agents for better results.
Contact dermatitis is the most reported side effect. A 2019 review in the International Journal of Cosmetic Science found that kojic acid at concentrations below 2% rarely caused skin sensitization, while concentrations above 4% increased the risk of irritation. It’s considered safe during pregnancy by most dermatologists, though less data exists compared to niacinamide or azelaic acid.
Alpha Arbutin
Safety rating: Safe. A milder alternative to hydroquinone.
Alpha arbutin is a naturally occurring derivative of hydroquinone found in bearberry, cranberry, and blueberry plants. It inhibits tyrosinase but releases hydroquinone at much lower levels than direct application, which accounts for its better safety profile.
The European Commission’s Scientific Committee on Consumer Safety (SCCS) evaluated alpha arbutin in 2015 and concluded it was safe for use in cosmetic face products at concentrations up to 2% and in body products up to 0.5%. Side effects are rare and limited to mild irritation in people with very sensitive skin.
Dangerous Ingredients to Avoid in Dark Spot Products
While most products from established brands are safe, the skin-lightening market has a serious problem with unregulated, imported products that contain genuinely dangerous substances.
Mercury
This is the biggest danger in unregulated skin-lightening products. The FDA has issued multiple warnings about mercury-containing creams sold in shops and online marketplaces. In a 2022 investigation by the Environmental Working Group (EWG), nearly half of 271 skin-lightening products tested contained mercury levels above 1 part per million (the legal limit). Some products from the Minnesota Department of Health’s testing contained mercury at levels as high as 54,000 ppm.
Mercury damages the nervous system, digestive system, immune system, lungs, kidneys, skin, and eyes, according to the FDA. Symptoms of mercury poisoning include tremors, memory problems, vision changes, depression, and numbness in the hands and feet. The danger extends beyond the user: family members can be exposed through contaminated towels, surfaces, and mercury vapors in the air.
Pregnant women and children are at the highest risk. Mercury passes through breast milk, and babies’ developing brains are especially vulnerable.
Red flags: Watch for products with handmade labels, labels in languages other than English, or missing ingredient lists entirely. Mercury may be listed as “mercurous chloride,” “calomel,” “mercuric,” “mercurio,” or simply “Hg.”
Unlisted Steroids (Corticosteroids)
Some imported skin-lightening products contain undisclosed corticosteroids like clobetasol propionate or betamethasone. These ingredients can temporarily lighten skin by reducing inflammation, but long-term use without medical supervision causes skin thinning (atrophy), stretch marks, increased infection risk, and potential adrenal suppression if absorbed systemically.
A 2020 study published in the British Journal of Dermatology found that 40% of unlabeled skin-lightening creams seized by UK Trading Standards contained potent corticosteroids.
High-Concentration Hydroquinone (Above 4%)
While prescription hydroquinone at 2-4% is used under medical supervision, unregulated products sometimes contain 6%, 8%, or even higher concentrations. At these levels, the risk of ochronosis, chemical burns, and permanent skin damage increases sharply. Never use hydroquinone products purchased from unregulated sources or without a valid prescription.
FDA vs. International Regulations on Skin Lightening Products
Regulations on dark spot removers vary significantly around the world, and understanding these differences matters if you purchase products online or while traveling.
- United States: OTC hydroquinone was removed from legal sale in 2020 under the CARES Act. Mercury is banned in cosmetics. The FDA regulates skin-lightening drugs but many imported products bypass enforcement.
- European Union: Hydroquinone is banned in all cosmetic products under EU Cosmetics Regulation (EC) No 1223/2009. It’s available only as a prescription drug. Mercury is also banned.
- Japan: Hydroquinone is allowed in cosmetics at concentrations up to 2%. Kojic acid is widely used and regulated as a quasi-drug ingredient.
- South Africa: Hydroquinone was banned in cosmetics in 1990, but enforcement has been inconsistent, and illegal products remain widely available according to a 2021 report from the South African Health Products Regulatory Authority (SAHPRA).
- India: Hydroquinone above 2% requires a prescription. The Drugs and Cosmetics Act regulates skin-lightening products, though enforcement varies by state.
The key takeaway: just because a product is legal or available in one country doesn’t mean it’s safe or legal in yours. Always check ingredient labels and buy from established retailers.
How to Patch Test Any Dark Spot Remover (Step-by-Step Protocol)
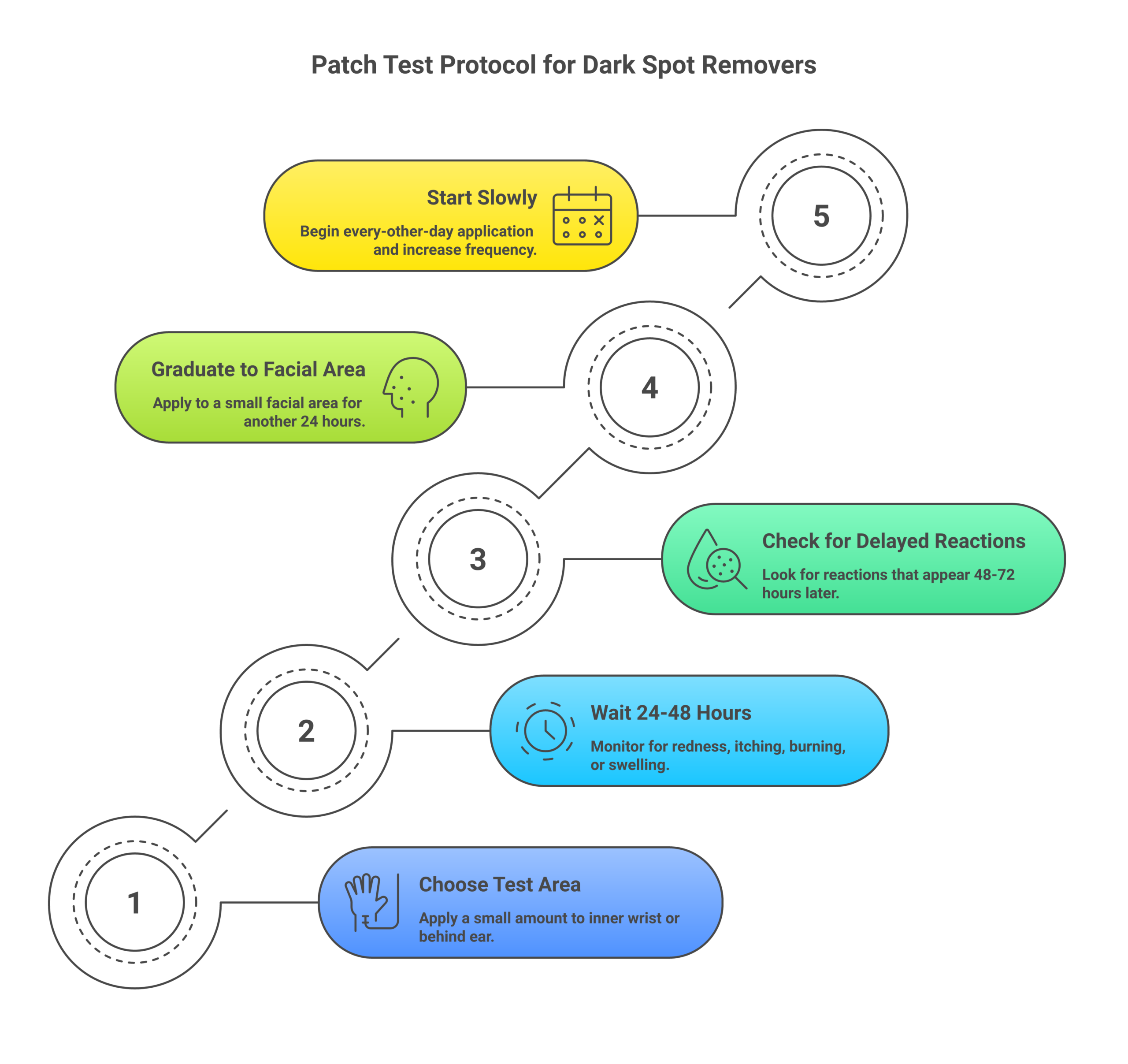
Patch testing is the single most effective way to identify a bad reaction before it affects your entire face. I recommend this protocol for every new product, no exceptions.
- Choose a test area: Apply a small amount of the product to the inside of your wrist or behind your ear. These areas have thinner skin that’s more likely to show a reaction.
- Wait 24-48 hours: Don’t wash the area. Monitor for redness, itching, burning, swelling, or any unusual changes.
- Check for delayed reactions: Some reactions, especially contact dermatitis from ingredients like kojic acid, can take 48-72 hours to appear.
- Graduate to a small facial area: If no reaction occurs, apply the product to a small area near your jawline for another 24 hours before using it on your full face.
- Start slowly: For active ingredients like retinoids or AHAs, begin with every-other-day application and increase frequency over 2-4 weeks as tolerated.
If you experience persistent burning, blistering, severe redness, or worsening dark spots at any stage, stop using the product immediately and consult a dermatologist.
When You Should See a Dermatologist
OTC products work well for mild hyperpigmentation from sun damage or minor acne scarring. But certain situations call for professional evaluation.
- Dark spots that change shape, color, or size: This could indicate melanoma or another skin condition that requires medical evaluation, not a dark spot remover.
- Melasma: This hormonal-driven hyperpigmentation is notoriously difficult to treat and often requires prescription-strength combinations (like Tri-Luma) plus strict sun avoidance.
- Dark spots after an inflammatory reaction: Post-inflammatory hyperpigmentation (PIH) sometimes needs prescription treatment, especially on darker skin tones where the risk of worsening pigmentation from aggressive OTC treatments is higher.
- No improvement after 8-12 weeks: If a product hasn’t shown visible results within this timeframe, it’s not working for you. A dermatologist can prescribe stronger options or combination therapies.
- Any adverse reaction: Blistering, chemical burns, persistent irritation, or paradoxical darkening all require professional attention.
Dark Spot Treatment Safety During Pregnancy and Nursing
Pregnancy changes the rules on dark spot treatment entirely. Hormonal shifts during pregnancy often trigger melasma (sometimes called the “mask of pregnancy”), but many of the most effective treatments are off-limits.
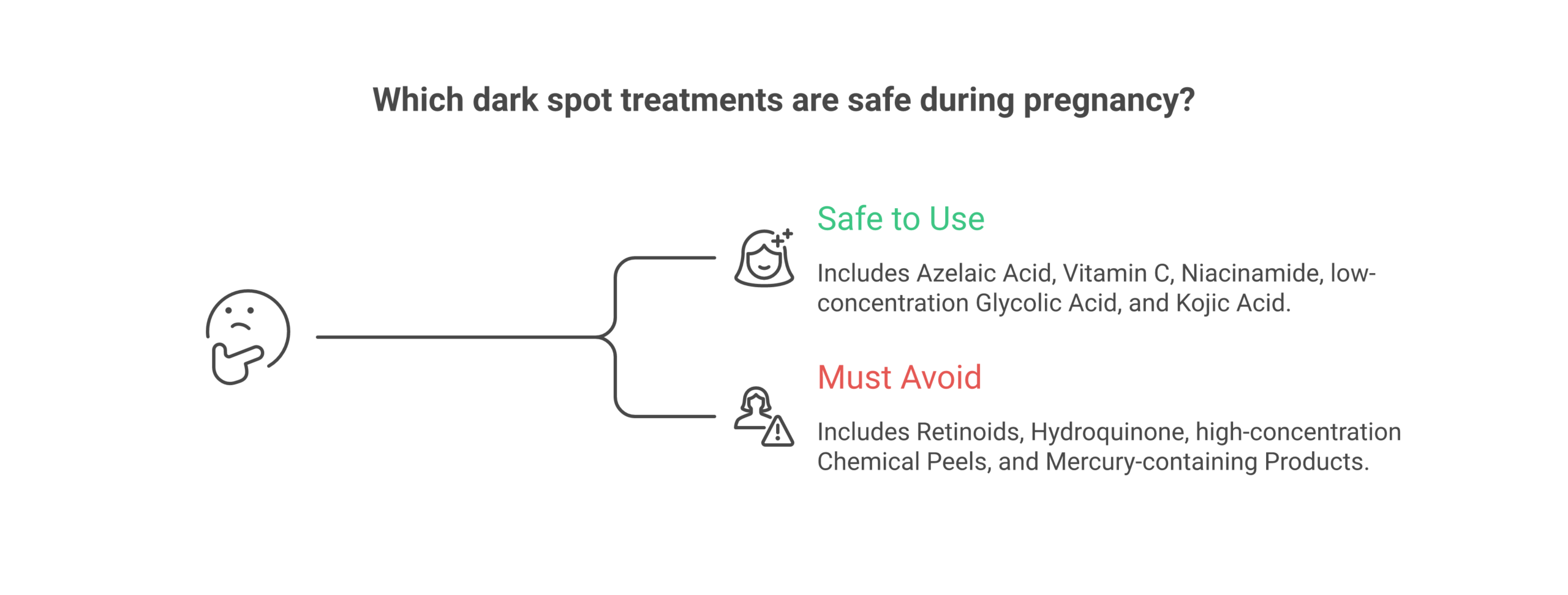
Safe During Pregnancy
- Azelaic acid (FDA Pregnancy Category B)
- Vitamin C (topical, any concentration used in cosmetics)
- Niacinamide (no known reproductive risks)
- Glycolic acid at low concentrations (below 10%, no professional peels)
- Kojic acid (generally considered safe, though less studied)
Avoid During Pregnancy
- All retinoids (retinol, tretinoin, adapalene, tazarotene): teratogenic, linked to birth defects
- Hydroquinone: limited safety data in pregnancy, 35-45% systemic absorption reported
- High-concentration chemical peels (above 10% glycolic acid or any TCA peels)
- Any product with mercury (mercury passes through breast milk and crosses the placenta)
The AAD recommends telling both your obstetrician and dermatologist about all topical products you use so they can adjust your skincare routine for pregnancy safety.
Long-Term Use: What Happens When You Use Dark Spot Removers for Months or Years
Many people use dark spot removers for extended periods because hyperpigmentation tends to recur without ongoing treatment. Here’s what the evidence says about long-term safety for each ingredient category.
- Vitamin C, niacinamide, alpha arbutin: No evidence of adverse effects with long-term daily use. These are suitable for indefinite use.
- AHAs: Safe for long-term use with consistent sunscreen application. Long-term exfoliation can thin the outer skin layer, so periodic breaks (1-2 weeks every 3-4 months) may help.
- Retinoids: Long-term use is well-documented and generally safe (outside of pregnancy). Some dermatologists recommend retinoids as lifelong skincare for anti-aging and pigmentation management.
- Hydroquinone: NOT safe for long-term use. Dermatologists typically limit use to 3-4 months on, then switch to maintenance with other ingredients. Extended continuous use increases ochronosis risk.
- Kojic acid: Generally safe for extended use at concentrations below 2%, though periodic skin monitoring is recommended.
Special Considerations for Melanin-Rich Skin (Fitzpatrick Types IV-VI)
If you have darker skin, you face a specific paradox with dark spot treatment: aggressive treatments designed to remove melanin can actually trigger more melanin production through post-inflammatory hyperpigmentation (PIH). This means the treatment itself can create new dark spots.
A 2022 systematic review published in the Journal of Cutaneous Medicine and Surgery found that treatment outcomes for PIH in skin of color showed “considerable room for improvement,” with many standard treatments carrying higher risk-to-benefit ratios for darker skin tones.
Here’s what’s safest for melanin-rich skin:
- Start with gentler ingredients: Niacinamide, azelaic acid, and mandelic acid (a larger-molecule AHA) are less likely to trigger PIH.
- Avoid aggressive peels: Professional chemical peels at high concentrations carry a significant PIH risk for Fitzpatrick types IV-VI.
- Use lower retinoid concentrations: Start at 0.025% tretinoin or a low-strength OTC retinol and increase gradually over months.
- Be cautious with lasers: Longer-wavelength lasers (1064 nm Nd:YAG) and picosecond lasers are considered safer for darker skin. Shorter-wavelength lasers can cause burns and worsen pigmentation.
- Sunscreen is non-negotiable: UV exposure worsens all forms of hyperpigmentation, and melanin-rich skin is not immune to UV-triggered dark spots despite higher baseline sun protection.
The Gut-Skin Connection and Dark Spot Prevention
At SANESolution, I’ve written extensively about how gut health affects skin health. Chronic inflammation driven by poor gut health can contribute to oxidative stress, which accelerates melanin overproduction. While dark spot removers treat the symptom, addressing gut health through anti-inflammatory nutrition, adequate sleep, and stress management supports the skin from the inside.
This doesn’t mean you should skip topical treatment. It means the safest, most effective approach combines a well-chosen dark spot remover with a foundation of whole-body health. The two strategies work better together than either one alone.
Quick Reference: Dark Spot Remover Safety Summary
| Ingredient | Safety Level | Pregnancy Safe? | Long-Term Use? | Key Risk |
|---|---|---|---|---|
| Vitamin C | Very Safe | Yes | Yes | Mild stinging at high concentrations |
| Niacinamide | Excellent | Yes | Yes | Rare irritation |
| Azelaic Acid | Very Safe | Yes (Cat. B) | Yes | Temporary tingling |
| Alpha Arbutin | Safe | Limited data | Yes | Mild irritation (rare) |
| Kojic Acid | Safe | Generally yes | Yes (<2%) | Contact dermatitis |
| AHAs | Safe with SPF | Low conc. only | Yes | Sun sensitivity (+18%) |
| Retinoids | Caution | NO | Yes | Teratogenic, irritation |
| Hydroquinone | Risk | NO | NO (<4 months) | Ochronosis, rebound |
Browse all of our dark spot removers reviews and guides for more research-backed recommendations.
If you are also looking for anti-aging solutions, see our guide to the best anti-wrinkle creams and learn about retinol for wrinkles.
Frequently Asked Questions About Dark Spot Remover Safety
Can dark spot removers make my skin worse?
Yes. Aggressive ingredients like high-concentration hydroquinone, strong retinoids, or chemical peels can trigger post-inflammatory hyperpigmentation, especially on darker skin tones. This is why patch testing and starting with lower concentrations matter. If your dark spots get darker or spread after starting a product, stop using it and see a dermatologist.
Are natural dark spot remedies (lemon juice, turmeric, apple cider vinegar) safe?
Not always. Lemon juice is highly acidic (pH 2) and can cause chemical burns, phytophotodermatitis (a severe skin reaction when combined with sun exposure), and worsen hyperpigmentation. Turmeric can temporarily stain the skin yellow but is generally harmless. Apple cider vinegar at full strength can damage the skin barrier. These DIY remedies lack the controlled concentrations and pH levels of formulated products, which is what makes them unpredictable.
How long does it take for a dark spot remover to work?
Most OTC dark spot removers take 8-12 weeks of consistent daily use to show visible results. Prescription-strength products may work faster (4-8 weeks). Any product claiming to remove dark spots in days is making a false claim. Melanin is produced deep in the epidermis, and it takes time for treated skin cells to migrate to the surface through natural cell turnover.
Is it safe to use multiple dark spot removers at the same time?
Some ingredient combinations work well together (vitamin C in the morning, retinol at night). Others can cause irritation or cancel each other out. Avoid combining AHAs with retinoids in the same application, as both are exfoliants and the combination can damage the skin barrier. Niacinamide pairs safely with almost everything. If you want to use multiple actives, introduce them one at a time with at least 2 weeks between each new product so you can identify which ingredient causes any reaction.
Do I need sunscreen while using a dark spot remover?
Yes, and this isn’t optional. UV exposure is the number-one cause of dark spots, and many active ingredients (retinoids, AHAs, hydroquinone, kojic acid) increase your skin’s sensitivity to UV damage. Using a dark spot remover without daily SPF 30+ sunscreen is counterproductive. You’re treating dark spots while creating the conditions for new ones. Apply sunscreen every morning, even on cloudy days, and reapply every 2 hours if you’re outdoors.
Are dark spot removers safe for all skin types?
Most dark spot ingredients are safe across skin types, but the approach should differ. Oily skin tolerates retinoids and AHAs well. Dry or sensitive skin benefits from gentler options like niacinamide and azelaic acid. Melanin-rich skin (Fitzpatrick IV-VI) needs extra caution because of the higher PIH risk from irritating ingredients. There’s no single product that’s ideal for everyone, which is why understanding your skin type and starting with a patch test matters.
What should I do if I have a bad reaction to a dark spot remover?
Stop using the product immediately. Wash the area with lukewarm water and a gentle cleanser. Apply a fragrance-free moisturizer to support the skin barrier. If symptoms are mild (redness, mild irritation), they should resolve within 24-48 hours. If you experience blistering, severe swelling, difficulty breathing, or worsening skin discoloration, seek medical attention right away. You can also report adverse reactions to the FDA’s MedWatch program at 1-800-FDA-1088.
Is hydroquinone still available anywhere in the U.S.?
Yes, but only by prescription. Since September 2020, OTC hydroquinone products are no longer legally sold in the United States. The only FDA-approved hydroquinone product is Tri-Luma, a prescription combination of hydroquinone 4%, tretinoin 0.05%, and fluocinolone acetonide 0.01%. Some compounding pharmacies also prepare custom hydroquinone formulations with a doctor’s prescription. If you find hydroquinone sold over the counter, it’s either leftover stock, being sold illegally, or an imported product that hasn’t gone through FDA review.
This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before starting any new skincare treatment, especially if you are pregnant, nursing, or have existing skin conditions.

